A new clinical trial in the Springfield area is giving cancer patients access to a potential way to reduce a common and often painful side effect of chemotherapy.
Mercy Health’s Springfield Cancer Center and Urbana Cancer Center are now participating in a study focused on chemotherapy-induced peripheral neuropathy, often called CIPN. That’s the nerve damage some patients experience during treatment, which can cause numbness, tingling, pain, and weakness—usually in the hands and feet.
For many people, those symptoms can make everyday life harder. Simple tasks like walking, working, or even holding objects can become difficult, and in some cases, the effects can last long after treatment ends.
“This is an exciting step forward in how we support patients through treatment,” said Lisa Gibson, Director of Oncology at Mercy Health – Springfield. She says the goal is not just to treat cancer, but to make treatment easier on patients and help them maintain their quality of life.
Right now, there aren’t many good options to prevent this kind of nerve damage during chemotherapy. That’s where this new study comes in.
The trial is testing a device called the Lilac Glove & Boot. It’s designed to gently apply pressure to a patient’s hands and feet during chemotherapy treatments. Researchers believe this may help limit how much chemotherapy reaches the nerves in those areas, which could reduce the risk of damage.
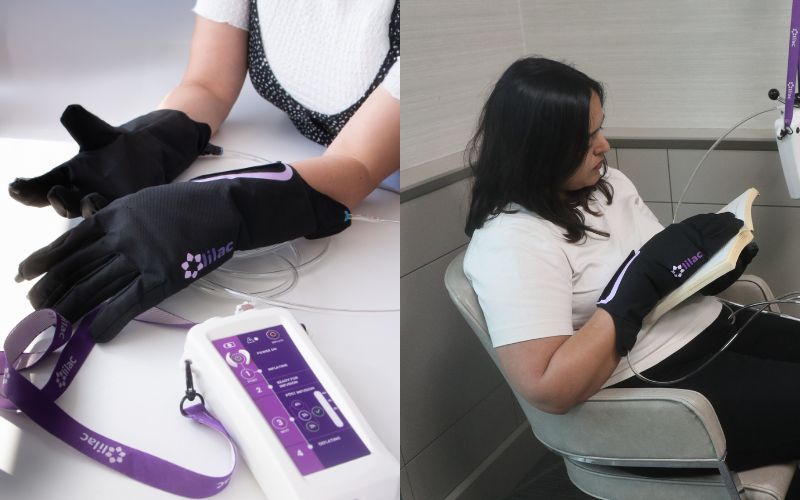
“It is easy to overlook how much we rely on our hands and feet until neuropathy starts to interfere with daily life,” said Oncology Clinical Research Nurse Chaundra Foss-Blizard. “Preserving that function during treatment could make a meaningful difference for patients.”
The study, known as the IMPACT clinical trial, is listed nationally as The Lilac Device Trial. It is being conducted with Luminate, the company that developed the device.
Lalita Kota, Director of Clinical Operations at Luminate, says many patients don’t realize how serious neuropathy can be until they experience it.
“Peripheral neuropathy is a devastating side effect of chemotherapy,” Kota said. “With no real solutions currently available to prevent it, we’re excited to bring this option to patients in Springfield.”
Patients who qualify for the trial will use either the active compression device or a comparison device during their treatments. Throughout the study, they’ll be closely monitored by their care team, tracking symptoms and progress.
In addition to receiving care, participants will also be helping researchers learn more about how to better support cancer patients in the future.
Mercy Health says this effort is part of a broader commitment to bringing advanced cancer care options closer to home. The Springfield Cancer Center has been serving patients for more than 20 years, while the Urbana Cancer Center expanded access to care when it opened last year.
Patients interested in the trial or wanting to learn more are encouraged to speak with their oncology care team.





